Peri-implantitis still remains one of the most common complications in implant dentistry. It is believed that peri‑implantitis is caused by bacterial‑driven inflammation around dental implants. Therefore, it has been suggested that administration of antibiotics, whether delivered systemically, locally, or via innovative carriers, can help target pathogens, thereby supporting decontamination and improving treatment outcomes for peri-implantitis. However, the effectiveness of antibiotic treatment for peri‑implantitis has still not been consistently demonstrated, and the studies show conflicting evidence for various treatment protocols.
New Positive Study: Use of Adjunctive Systemic Azithromycin to Treat Peri-implantitis
| Study design | How azithromycin was used | Main findings |
|---|---|---|
| Retrospective, non‑surgical (submucosal debridement + chlorhexidine) | 500 mg /day for 3 days | After a mean 22.9 months, probing pocket depth fell ≈ 3.9 mm, intrabony defects filled ≈ 2.6 mm, and disease resolution occurred in 66.7 % of implants. 1 |
Within the limitations of the study design, non-surgical approach with adjunctive use of systemic azithromycin for the treatment of peri-implantitis associated lesions with intrabony defects may lead to significant clinical and radiographic improvements. 1
Prior Studies were Inconclusive or Showed No Clear advantage in Some Surgical Cases
Below are two other recent studies, using adjunctive systemic antibiotic in the treatment of peri‑implantitis. The evidence of its benefit is mixed.
| Study design | How azithromycin was used | Main findings |
|---|---|---|
| Previous Studies | ||
| Retrospective, flap‑less surgical (curettage ± prosthesis modification)3 | One hundred and seventeen patients with 338 implants were included. Systemic azithromycin (or metronidazole) given to some patients | Use of systemic azithromycin was identified as a positive predictor for disease resolution; overall, 54.4 % of implants achieved resolution. |
| A total of 52 patients with stage III/IV grade C periodontitis were randomly allocated to two groups receiving NSI with or without adjunctive azithromycin. 4 | Use of oral azithromycin as an adjunct to non-surgical subgingival instrumentation | No significant differences were observed in the levels of periodontal pathogens at 12 months except for Prevotella intermedia. No significant differences were observed for the studied cytokines at 3 and 12 months. |
Case Photos: Peri-implantitis Overview
Case photos below provided by: Rokaya D, Srimaneepong V, Wisitrasameewon W, Humagain M, Thunyakitpisal P. Peri-implantitis Update: Risk Indicators, Diagnosis, and Treatment
Peri-implant mucositis inflammation
Peri-implant mucositis demonstrating soft-tissue inflammation.
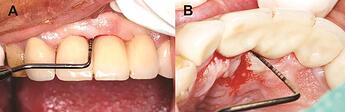
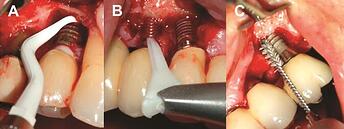
Peri-implantitis Clinical Manifestation
Peri-implantitis: ( A ) clinical photograph, ( B ) radiograph showing bone loss around an implant, and ( C ) clinical photograph showing buccal bone loss.
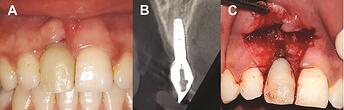
Retained cement results in peri-implantitis:
Retained cement results in peri-implantitis: ( A ) retained cement at the crown margin and excess cement in the peri-implant inflamed tissue, and ( B ) excess cement around the abutment. Reproduced from Ref. 23 with permission from John Wiley & Sons A/S.
Various treatments for peri-implant diseases.
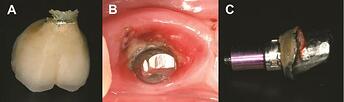
Mechanical treatments of peri-implantitis:
Mechanical treatments of peri-implantitis: ( A ) plastic curettes, ( B ) air abrasive, and ( C ) metallic brush. Reproduced from Ref. 34 under the Creative Commons Attribution License (CC BY 4.0) from Frontiers Media S.A.
Case: Simplified nonsurgical treatment of peri-implantitis using chlorhexidine and minocycline hydrochloride
Case photos below provided by: Heo S, Kim HJ, Joo JY, Lee J, Kim SJ, Choi J. Simplified nonsurgical treatment of peri-implantitis using chlorhexidine and minocycline hydrochloride.
Forty-five dental implants in 20 patients diagnosed with peri-implantitis were included. At baseline and the study endpoint, the probing pocket depth (PPD), clinical attachment level (CAL), and the presence of bleeding on probing (BOP) at 6 sites around each implant were recorded. The radiographic osseous defect morphology at the mesial or distal proximal aspect of each implant was classified as 1) narrow or wide and 2) shallow or deep. For a comparative analysis of bone changes according to the defect morphology, the distance from the implant shoulder to the most coronal bone-to-implant contact point (DIB) at the mesial and distal aspects of each implant was measured at baseline and the endpoint. Patients were scheduled to visit the clinic every 2–4 weeks for intrasulcular irrigation of chlorhexidine and delivery of minocycline HCl.
We observed statistically significant decreases in PPD, CAL, and BOP after treatment. At the endpoint, bone levels increased in all defects, regardless of the osseous morphology of the intrabony defect. The mean DIB change in deep defects was significantly greater than that in shallow defects. Although the mean bone gain in narrow defects was greater than in wide defects, the difference was not statistically significant.
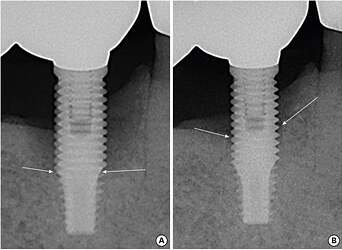
Figure 1. A representative radiographic view at the baseline day (A) and the endpoint (B).
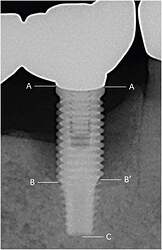
Figure 2. The distance from the implant shoulder to the most coronal bone-to-implant contact point (DIB) was measured at the mesial and distal aspects of each implant: A, implant shoulder; B and B′, the most coronal bone-to-implant contact point; C, apex of the implant fixture; A–B, DIB at the distal aspect; A–B′, DIB at the mesial aspect; and A–C, length of the implant fixture (on radiographic view). Amount of actual marginal bone loss at the mesial and distal aspects (X′ and X) calculated follow as:
X′=(A–B′)×fixture length (actual)/(A–C) X=(A–B)×fixture length (actual)/(A–C)
References
-
Leira Y, Manfredi G, Tortosa-Álvarez F, González-Moure A, Rodríguez-Díaz B, Vázquez-Reza M, Vázquez-Taín F, Liñares A, Guerrero A, Blanco J. Non-Surgical Treatment of Peri-implantitis Associated Intrabony Defects with the Use of Adjunctive Systemic Azithromycin: Clinical and Radiographic Outcomes of a Dual-Centre Retrospective Study. Int J Oral Maxillofac Implants. 2026 Mar 6;0(0):1-24.
-
Rokaya D, Srimaneepong V, Wisitrasameewon W, Humagain M, Thunyakitpisal P. Peri-implantitis Update: Risk Indicators, Diagnosis, and Treatment. Eur J Dent. 2020 Oct;14(4):672-682. doi: 10.1055/s-0040-1715779.
-
Carrillo de Albornoz A, Montero E, Alonso-Español A, Sanz M, Sanz-Sánchez I. Treatment of peri-implantitis with a flapless surgical access combined with implant surface decontamination and adjunctive systemic antibiotics: A retrospective case series study. J Clin Periodontol. 2024 Aug;51(8):968-980.
-
Sulugodu Ramachandra S, Woodford V, Han P, Lee RSB, Ivanovski S. Systemic Azithromycin as an Adjunct to Non-Surgical Subgingival Instrumentation in the Treatment of Stage III/IV, Grade C Periodontitis: 12-Month Clinical, Microbiological and Cytokine Results of a Randomised Controlled Trial. J Clin Periodontol. 2025 May;52(5):666-680.
-
Heo S, Kim HJ, Joo JY, Lee J, Kim SJ, Choi J. Simplified nonsurgical treatment of peri-implantitis using chlorhexidine and minocycline hydrochloride. J Periodontal Implant Sci. 2018 Oct 29;48(5):326-333.
-
Tord Berglundh,Andrea Mombelli,Frank Schwarz,Jan Derks et al., Periodontology 2000. Volume: , Issue: , 2024 Etiology, pathogenesis and treatment of peri-implantitis: A European perspective.
Peri-implantitis is a plaque-associated pathological condition occurring in tissues around dental implants. It is characterized by inflammation in …
- Xiaoyan Li,Ailin Wei,Haosen Zhao,Zhenfei Wang,Yang Lyu,Jie Nie,Yu Chen et al., Colloids and surfaces. B, Biointerfaces. Volume: 242, Issue: , 2024
A carboxymethyl-resistant starch/polyacrylic acid semi-IPN hydrogel with excellent adhesive and antibacterial properties for peri-implantitis prevention.
Hydrogels possess inherent characteristics that render them promising for the prevention of peri-implantitis. Nonetheless, hydrogels with singular …
-
Manuel Toledano,María T Osorio,Marta Vallecillo-Rivas,Manuel Toledano-Osorio,Alberto Rodríguez-Archilla,Raquel Toledano,Raquel Osorio et al., Journal of dentistry. Volume: 113, Issue: , 2021
Efficacy of local antibiotic therapy in the treatment of peri-implantitis: A systematic review and meta-analysis.
-
Lisa J A Heitz-Mayfield,Giovanni E Salvi,Andrea Mombelli,Pierre-Jean Loup,Fritz Heitz,Estie Kruger,Niklaus P Lang et al., Clinical oral implants research. Volume: 29, Issue: 1, 2018
Supportive peri-implant therapy following anti-infective surgical peri-implantitis treatment: 5-year survival and success.
To evaluate clinical outcomes of supportive peri-implant therapy (SPIT) following surgical treatment of peri-implantitis. :…