This Week in Dental Implants we wanted to highlight an excellent article and case series that reviews the techniques and applications of L-PRF for ora-antral communications, sinus lifts, socket preservation, and mandibular third molar surgery.
What is L-PRF?
L-PRF, leukocyte and platelet-rich fibrin, is just one of the many flavors of PRF. It simply represents a solid PRF protocol (creation of PRF plugs and membranes, as opposed to liquid), similar to advanced platelet-rich fibrin (A-PRF), but is differentiated from A-PRF by it’s higher Relative Centrifugal Force (RCF), or g-force. For lack of better terms, A-PRF is a low-centrifugation protocol for slightly longer period of time, while L-PRF is a high-centrifugation protocol for a shorter period of time. See the table before more details.
![]() Be careful with blind reliance on RPM Values
Be careful with blind reliance on RPM Values
Most of the literature, including the study referenced below, uses the centrifuge RPM (rotation per minute) to describe the particular PRF protocols. This is very confusing as the RPM, simply measures how fast the rotor is spinning, and does not provide a true measure of the force the same is subject to. The proper RPM for any PRF protocol, will therefore, be different depending on the centrifuge used. Given the wide range of centrifuges and types, both fixed-angle and horizontal, you cannot simply use the RPM referenced in a study on the particular machine being used, unless you are certain you are using the same exact centrifuge as that employed in the study. As such, it is vital to always translate the RPM into a Relative Centrifugal Force (RCF), or g-force. RCF accommodates the rotor size into its measurement so you can easily can determine the gravitational force being applied. RCF can then be used as a common value for any centrifuge.
Sample RPM Protocol for different platelet concentrates (1,2)
![]() IMPORTANT
IMPORTANT
As noted above, the RPM to be used for each version of PRF will be DIFFERENT depending on a particular centrifuge. The RPM values below are those referenced in this study and which was done on a fixed angle centrifuge. However, from our research (and we maybe incorrect about this), the original L-PRF protocol was ~700 RCF max for 12 minutes, while A-PRF was approximately ~200 RCF max for 14 minutes (low centrifugation protocol) or more recently 8 minutes (A-PRF +), see: Technical considerations in obtaining platelet rich fibrin for clinical and periodontal research for a great overview.
| Platelet concentrate | Fixed Angle Protocol (rpm/min) | Presentation form |
|---|---|---|
| L-PRF | 3,000/12 (some machines at 2,700) | Solid |
| A-PRF | 1,500/14 (A-PRF+ 8 min) | Solid |
(L-PRF: leukocyte platelet-rich fibrin, A-PRF: advanced platelet-rich fibrin)
Application Summary of leukocyte‑platelet‑rich fibrin (L‑PRF) in oral and maxillofacial surgery
| Patient / Situation | L‑PRF Use | Outcome |
|---|---|---|
| Patient 1 – oroantral communication | L‑PRF membrane + vestibular flap | Successful closure and satisfactory soft‑tissue healing |
| Patient 2 – maxillary sinus lift | L‑PRF membranes placed over bone graft (replacing collagen membranes) | Reduced cost and enhanced bone regeneration |
| Patients 3‑5 – systemically compromised, post‑extraction | L‑PRF plug and membranes inserted into extraction sockets | Prevented complications, preserved alveolar ridge for future rehabilitation |
| Patients 6‑7 – recurrent pericoronitis of partially erupted lower third molars | Extraction followed by L‑PRF plug and membrane placement | Uneventful healing in all cases |
Overall conclusion
L‑PRF proved to be a versatile, biocompatible autologous material that supports soft‑tissue closure, promotes bone regeneration (e.g., sinus lifts, ridge preservation), and facilitates uneventful healing after extractions and other oral‑maxillofacial procedures.
Case Presentations
![]() The Case photos below are Seixas DR, Sanches IM, Mazzo A, Santos PSDS, Gonçales ES. Applications of leukocyte platelet-rich fibrin in oral and maxillofacial surgery: case series.
The Case photos below are Seixas DR, Sanches IM, Mazzo A, Santos PSDS, Gonçales ES. Applications of leukocyte platelet-rich fibrin in oral and maxillofacial surgery: case series.
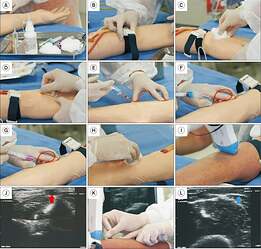
Venipuncture and blood collection.
Venipuncture and blood collection. A. Materials required for venipuncture: gauze or hydrophilic cotton, antiseptic solution, disposable scalpel or disposable needle, needle adapter, vacuum blood collection tubes, tube rack, sterile dressings, and a rigid sharps container. In addition, the clinician must wear all appropriate PPE. B. Tourniquet in place (5-10 cm above the puncture site). C. Antisepsis of the puncture site. D. Needle insertion. E. Blood collection in the first vial. F. The first vial was removed while keeping the needle in position. G. Insertion of the second vial to continue blood collection. H. Compression of the puncture site with gauze after needle removal. I. In cases of difficulty locating the vein, a portable ultrasound may assist in identifying the optimal puncture site. J. Ultrasound image showing the target vein for puncture. The red arrow highlights the vein selected for venipuncture. K. Ultrasound guidance during needle insertion. L. Ultrasound image showing the needle inside the vessel (blue arrow).
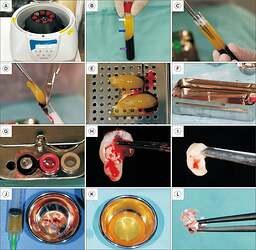
Protocol for the preparation of leukocyte- and platelet-rich fibrin (L-PRF).
Protocol for the preparation of leukocyte- and platelet-rich fibrin (L-PRF). A. Blood tubes with red caps are centrifuged at 3,000 rpm for 12 minutes to prepare L-PRF. B. After centrifugation, three layers resulting from blood cell separation become visible: a top layer of platelet-poor plasma (white arrow), a middle layer consisting of the L-PRF clot (blue arrow), and a bottom layer of red blood cells (purple arrow). C. Using sterile long tweezers, the clinician carefully removes the L-PRF clot from the tube. D. The L-PRF is gently separated from the red blood cell layer with the aid of a sterile scalpel or scissors. E. The clots are then placed on the L-PRF processing box (e.g., PRF box or PRF kit). F. To fabricate an L-PRF membrane, the clots are covered with the compression plate, which applies constant and uniform pressure. G. To fabricate L-PRF plugs, the clots are inserted into the cylindrical wells of the PRF box. H. L-PRF membrane after 5 minutes of compression. I. L-PRF plug after 5 minutes of compression. J. Using this protocol, the clinician can aspirate the exudate (expressed serum) to be used in combination with the L-PRF clots. K. By applying a different centrifugation protocol (3,300 rpm for 2 minutes), Injectable platelet-rich fibrin (i-PRF) can be obtained. L. i-PRF can be mixed with bone graft materials to produce “sticky bone”.
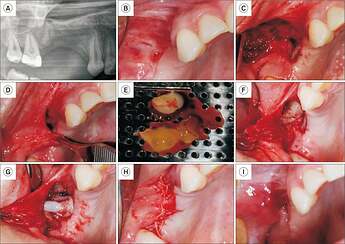
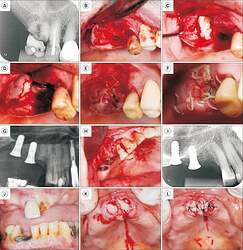
Closure of oroantral communication with leukocyte platelet-rich fibrin (L-PRF).
Closure of oroantral communication with leukocyte platelet-rich fibrin (L-PRF). A. Initial radiograph showing oroantral communication in the posterior maxillary region on the right side. B. Initial clinical appearance showing small fistulas in the vestibular region of the maxilla. C. Appearance after the creation of a vestibular flap. D. Vestibular flap after dissection, highlighting that it is possible to pull it without tensioning the region. E. L-PRF membranes were obtained by centrifugation at 3,000 rpm for 12 minutes. F. First L-PRF membrane in position. G. Second L-PRF membrane in position. H. Suture of the region. I. Clinical appearance 15 days after surgery.
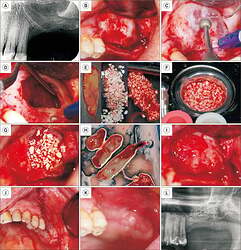
Sinus lift with leukocyte platelet-rich fibrin (L-PRF).
Sinus lift with leukocyte platelet-rich fibrin (L-PRF). A. Periapical radiograph of the left upper molar region showing extensive pneumatization of the left maxillary sinus. B. Access to the lateral wall of the maxillary sinus. C. Creation of the lateral bone cavity using a round bur. D. Detachment and superior elevation of the Schneiderian membrane using a curette. E. Placement of an L-PRF membrane combined with biomaterial particles adjacent to the Schneiderian membrane. F. Preparation of sticky bone from a mixture of biomaterial and injectable injectable platelet-rich fibrin (i-PRF). G. Positioning of the sticky bone. H. L-PRF membranes were obtained by centrifuging blood at 3,000 rpm for 12 minutes. I. L-PRF membranes covering the sticky bone. J. Flap positioned and secured with sutures. K. Clinical appearance 15 days post-surgery. L. Radiographic examination 15 days post-surgery.
Bone socket preservation with leukocyte platelet-rich fibrin (L-PRF).
Bone socket preservation with leukocyte platelet-rich fibrin (L-PRF). A. Initial periapical radiograph of Patient 3, showing extensive coronal destruction of tooth 16. B. Following extraction, a sizable purulent collection was observed in the distal root area, which was curetted. C. An L-PRF plug and membrane were placed in the area that was curetted. D. An L-PRF plug and membrane were also placed in the mesial root area. E. Immediate suture. F. Clinical appearance 15 days post-extraction. G. Initial radiograph of Patient 4, showing a root remnant in the region of tooth 15, adjacent to implants. H. After root extraction, L-PRF membranes were positioned both in the alveolar area and around the neighboring implant. I. Immediate periapical radiograph post-extraction. J. Patient 5, with a history of cancer treatment, presenting teeth 11 and 21 indicated for extraction. K. L-PRF plugs positioned in each socket. L. L-PRF membrane sutured over the sockets. Note that exposure of the L-PRF membrane in the oral cavity does not pose any issues.
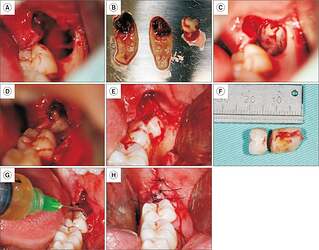
Mandibular third molar surgery with leukocyte platelet-rich fibrin (L-PRF).
Mandibular third molar surgery with leukocyte platelet-rich fibrin (L-PRF). A. Post-extraction socket of tooth 38. B. L-PRF membranes and plug prepared. C. L-PRF plug positioned within the extraction socket. D. L-PRF membranes placed over the plug and beneath the flap. E. Impacted third molar exposed after access via envelope flap. F. Extracted third molar. G. Irrigation of the socket with injectable platelet-rich fibrin (i-PRF). H. Sutures in place.
References
- Seixas DR, Sanches IM, Mazzo A, Santos PSDS, Gonçales ES. Applications of leukocyte platelet-rich fibrin in oral and maxillofacial surgery: case series. J Korean Assoc Oral Maxillofac Surg. 2025 Dec 31;51(6):384-396.
- Jamalpour MR, Shahabi S, Baghestani M, Shokri A, Jamshidi S, Khazaei S. Complementarity of surgical therapy, photobiomodulation, A-PRF and L-PRF for management of medication-related osteonecrosis of the jaw (MRONJ): an animal study. BMC Oral Health. 2022 Jun 18;22(1):241.
- Bains VK, Mahendra J, Mittal M, Bedi M, Mahendra L. Technical considerations in obtaining platelet rich fibrin for clinical and periodontal research. J Oral Biol Craniofac Res. 2023 Nov-Dec;13(6):714-719.